| Home | Phyllosphere | Culturing | Jellies | Molecular Methods | Publications |
|---|
Discovering the Diet of the Phyllosoma

Much remains to be understood about the ecology of spiny lobsters (rock lobsters), especially during their larval stages. Their larval diet has eluded discovery for some time, despite it being crucial to understanding this part of their life-history.
Phyllosomas migrate hundreds of kilometers offshore into nutrient poor waters, where they feed. This offshore phase can last for up to nine months for the Western Australian rocklobster and as long as two years for the New Zealand species. They eventually moult into a non-feeding post-larvae and swim across the shelf break. Understanding the phyllosomas’ food-web, which ultimately fuels this onshore migration, is therefore crucial to understanding post-larval recruitment and the probability that lobster populations will endure. Understanding diet could also facilitate culture of larval stages. Determining the diet of spiny lobster larvae was difficult before molecular techniques; these animals are paper thin and transparent (and the prey is mushed up and transparent too). They are also patchily distributed and occur hundreds (and up to thousands) of kilometers offshore, which makes direct observation of feeding almost impossible.
This research was mostly conducted as part of a PhD with Associate Professor Shane Lavery and Professor Andrew Jeffs.
Diet of phyllosomas of Aotearoa New Zealand
We collected samples on six oceanic voyages lead by Scott Nodder of NIWA. The voyages were to recover data from two moorings located on the sub-Antarctic front and just North of the Chatham rise. The Northern mooring was near a large patch of ocean in which lobster larvae occur, and Scott was exceptionally generous and let us do research during the night after they had collected their mooring and were cleaning it down and recovering data before redeploying it in the morning.
Lobster larvae occur at pretty low density. On the first voyage we towed a plankton net (1.6 x 1.6 m opening) and got one single red rock lobster larva (Jasus edwardsii) and on the second voyage we got three more larvae with the same net over six hours, and also two slipper lobster larvae. Simon Connell used taxon specific PCR to see whether jellyfish, arrow worms or fish were in the guts of these. He found that three were positive for cnidarian (jellyfish), two contained fish and none contained arrow worm DNA.
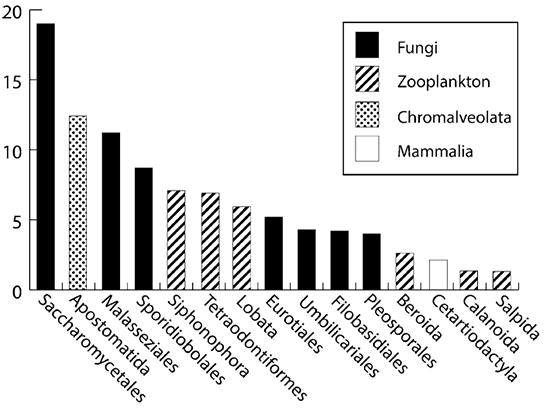

Dr Miao Wang and I collected a further fifteen larvae on subsequent voyages and we used 454 amplicon sequencing to detect prey. For this study we used “universal” primers that would amplify most Eukaryotes. The advantage of this approach is that we don’t need to make any assumptions about what is in the larval guts. Surprisingly the most larvae were full of fungi (predominantly yeasts). We also detected quite a few microscopic Eukaryotes that are typically associated with crustaceans. The prey items we found were: fish (are these larvae, eggs?) and gelatinous animals such as siphonophores, comb jellies and salps.
The western rock lobster Panulirus cygnus
The western rocklobster fishery is remarkable because it has been sustainably managed for many decades, but has historically made an annual landed profit of around half a billion dollars. Scientific measurement and modeling have been key to this successful marriage of resource management and industry. Consequently, some of the earliest and most thorough insights into the ecology of spiny lobster larvae are of the western rocklobster in the East Indian Ocean.
We sucked the mid-gut contents from wild larvae and then sequenced two short regions of the gene for the 18S subunit of the nuclear ribosome. By sequencing two linked loci we could confirm that there wasn’t too much distortion of results from PCR (which can be a big problem with low copy and degraded DNA, such as you get from the gut). Surprisingly, there was a considerable quantity of DNA from colonial radiolarians. These large gelatinous colonies break up when sampled by plankton net, which means that they can be overlooked, but video plankton recorders have shown them to be a substantial component of the plankton around Australia. Without using an eDNA approach I doubt that these organisms would have been considered a viable prey. We also found gelatinous zooplankton such as siphonophores, scyphozoans (true jellyfish) and salps to be a part of the diet.
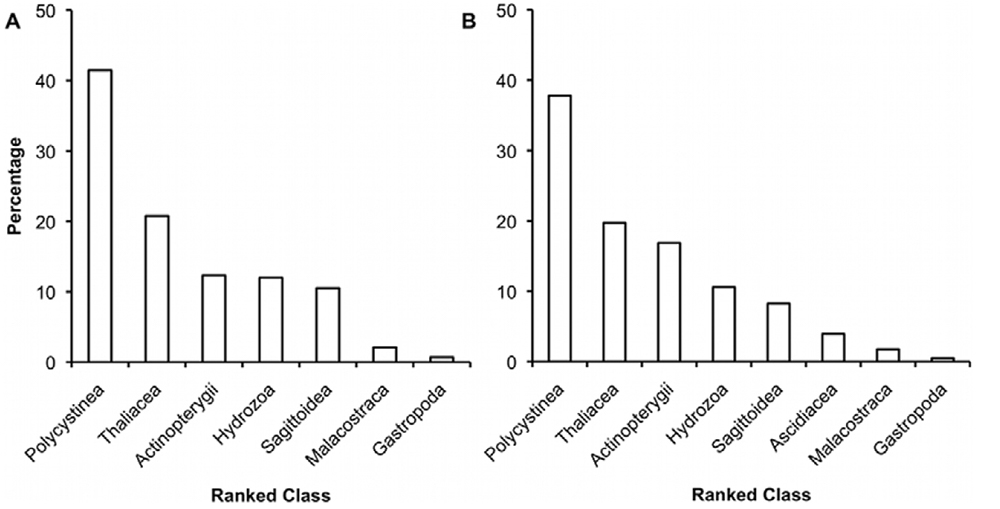
We returned to the East Indian Ocean the following year to collect more samples and drill a little deeper into how the ecology of the East Indian ocean impacts on lobster larvae. The East Indian Ocean consists of a field of counter-rotating eddy pairs, with each eddy being around 200 km in diameter (mesoscale eddies). We sampled two pairs of eddies and collected numerous larvae in a range of mid to late developmental states. Most larvae were in stages VI, VII and VIII, but we also collected a few that were stages V and IX (there are nine developmental stages). This meant that we had enough samples to meaningfully compare the diet and health of three larval stages of larvae across two clockwise and two anticlockwise rotating eddies.
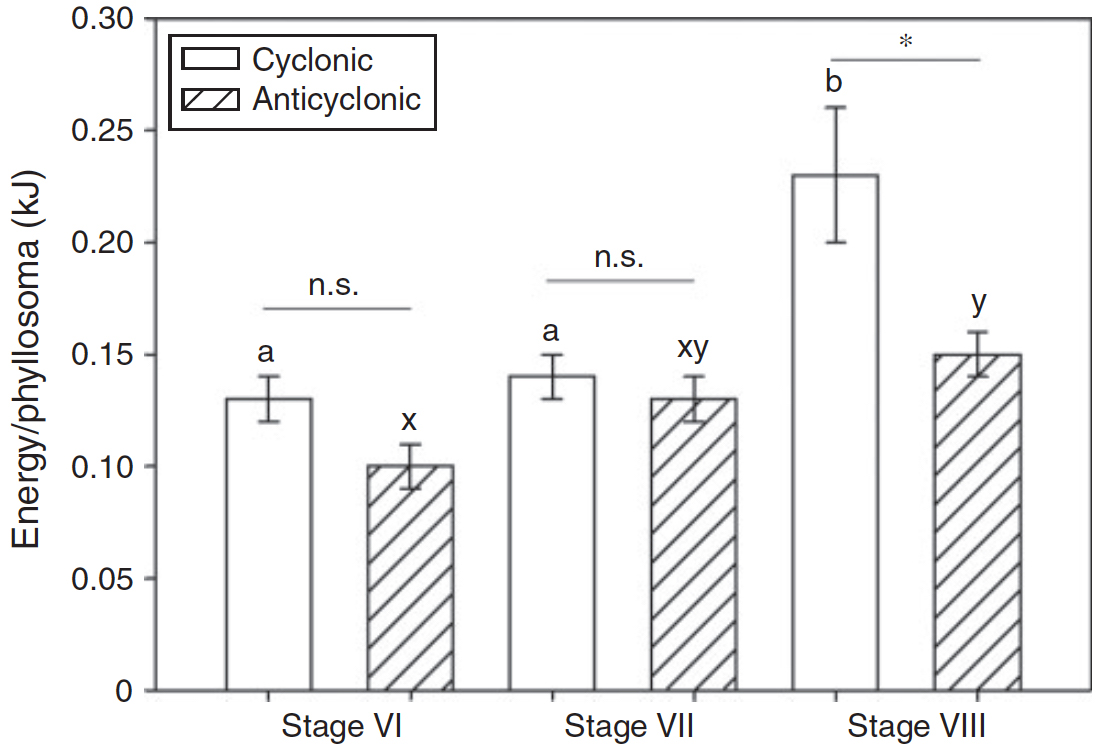
The most important discovery was made by Dr Miao Wang, who found that the larvae taken from anticlockwise eddies were considerably (and significantly) smaller than those in clockwise eddies. This should impact on the number of larvae that are able to make it back onshore and develop into adult lobsters, because the larger larvae carry more energy to fuel the animals’ active migration back over the shelf break. Also, the lipid reserves were on average 67% less and protein levels 39% less in developmental stage VIII phyllosomas. This result was surprising because phyllosomas were almost twice as abundant in anticlockwise eddies in 2011 - and anticlockwise eddies (that in the Southern hemisphere have a deeper warm core) have consistently been shown to be more productive overall and to typically have richer prey-fields than their clockwise rotating counterparts.
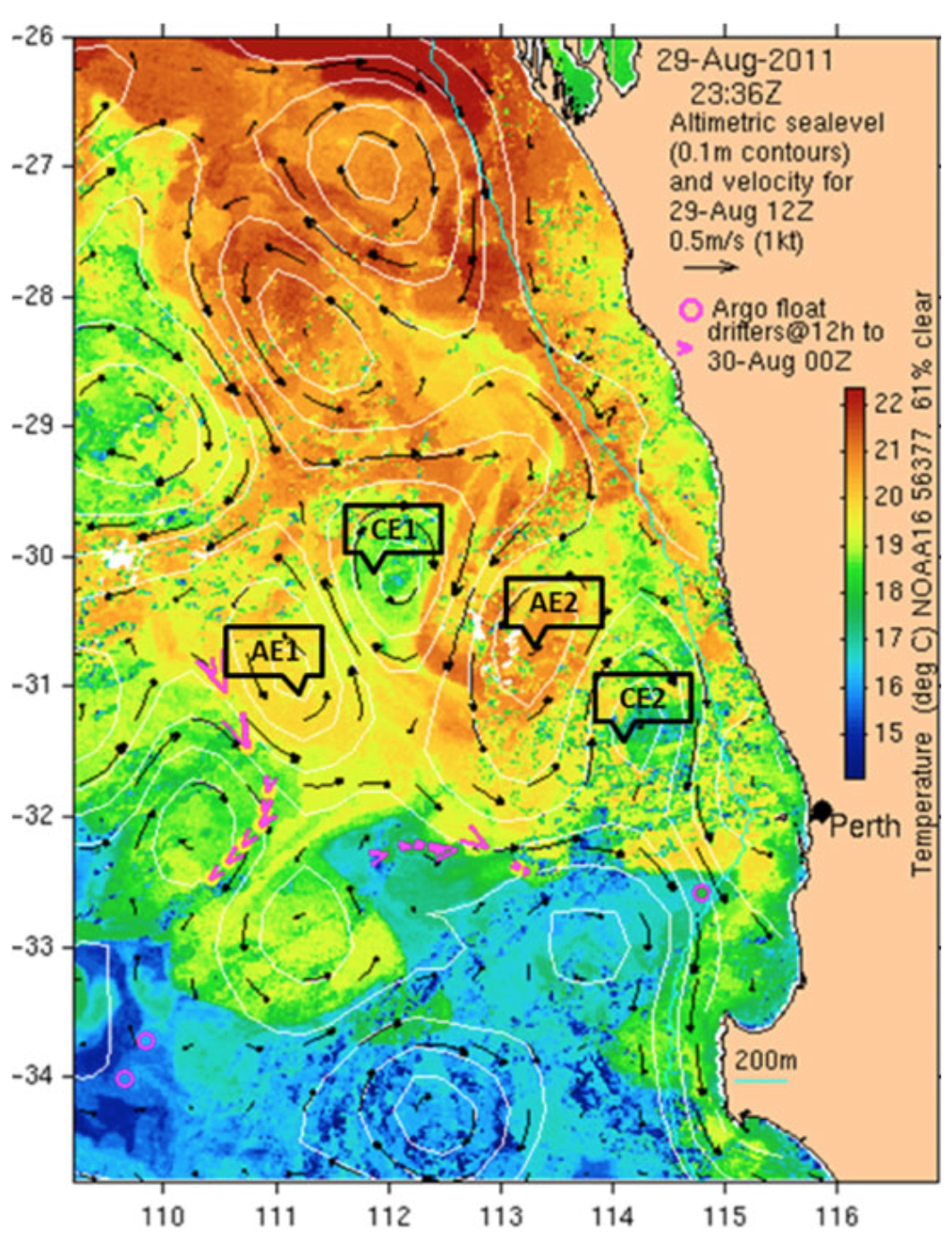
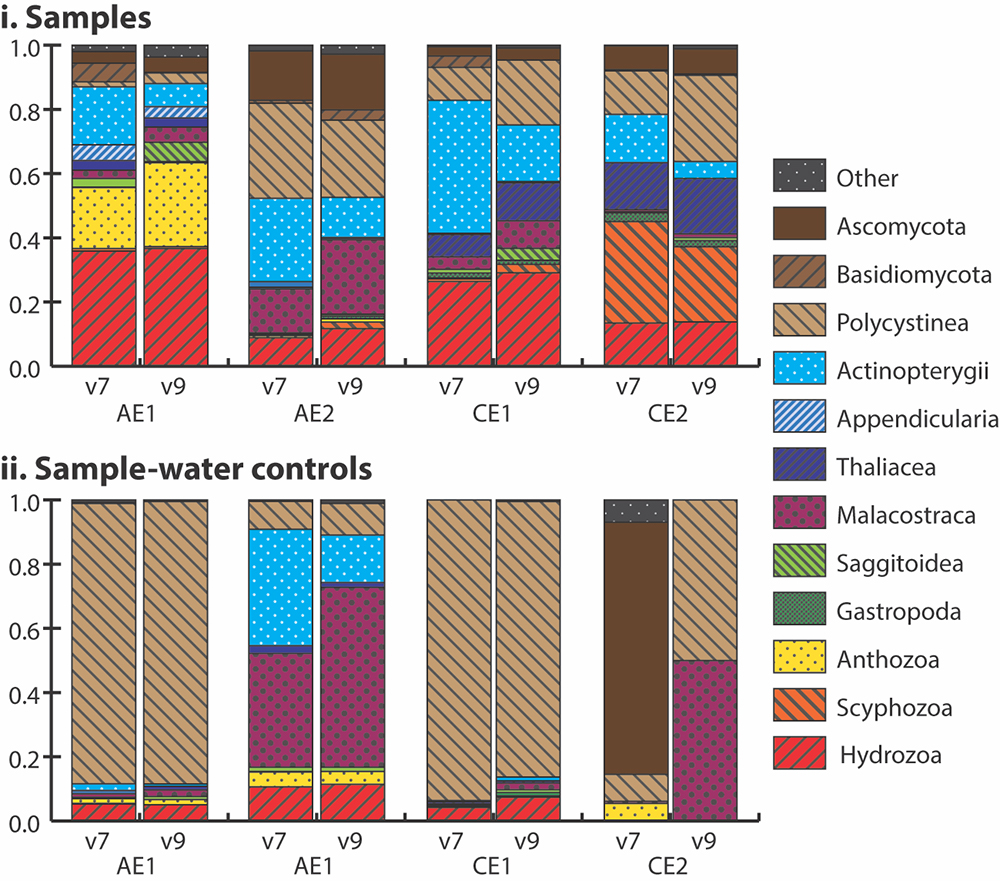
We sequenced the partly digested prey DNA from the guts of many free-floating lobster larvae of developmental stages VI, VII and VIII that were sampled from the counter-rotating eddies. Although there were small differences in the prey species detected in the larvae from different eddies, none of these could be meaningfully associated with the direction that the eddy was rotating. This suggests that the varying condition of larvae sampled from counter-rotating eddies was not due to the composition of their immediate diet. This suggests that the explanation for the difference in larval condition across eddies is either not dietary, or if it is dietary it could be due to absolute (rather than relative) abundances of their favored prey or that the dietary impacts on larval health occurred before the time of sampling. Dietary markers in the tissues of the larvae did indicate that there was a difference in the food-web composition of the counter-rotating eddies at a more fundamental level.
Read the Publications
O’Rorke, R., S.D Lavery, M. Wang, R. Gallego, A.M. Waite, L.E. Beckley, P.A. Thompson, and A.G. Jeffs. (2014) Phyllosomata associated with large gelatinous zooplankton: hitching rides and stealing bites. ICES Journal of Marine Science doi:10.1093/icesjms/fsu163
O’Rorke, R., Lavery, S. D., Wang, M., Nodder, S. D., and Jeffs, A. G. (2013). Determining the diet of larvae of the red rock lobster (Jasus edwardsii) using high-throughput DNA sequencing techniques. Marine Biology doi:10.1007/s00227-013-2357-7
Wang, M., O’Rorke, R., Nodder, S., Jeffs, A. (2014) Nutritional composition of potential prey of the spiny lobster phyllosoma (Jasus edwardsii). Marine and Freshwater Research 65, 3337-349
Wang, M., O’Rorke, R., Waite, A. M., Beckley, L. E., Thompson, P. A., and Jeffs, A.G. (2014). Fatty acid profiles of phyllosoma larvae of western rock lobster (Panulirus cygnus) in cyclonic and anticyclonic eddies of the Leeuwin Current off Western Australia. Progress in Oceanography, 122, 153–162
O’Rorke, R., Lavery, S. D., Wang, M., Nodder, S. D., and Jeffs, A. G. (2013). Determining the diet of larvae of the red rock lobster (Jasus edwardsii) using high-throughput DNA sequencing techniques. Marine Biology doi:10.1007/s00227-013-2357-7
O’Rorke, R., Jeffs AG, Fitzgibbon Q, Chow S, Lavery S (2013) Extracting DNA from whole organism homogenates and the risk of false positives in PCR based diet studies; a case study using spiny lobster larvae. Journal of Experimental Marine Biology and Ecology 441: 1-6
| Home | Phyllosphere | Culturing | Jellies | Molecular Methods | Publications |
|---|